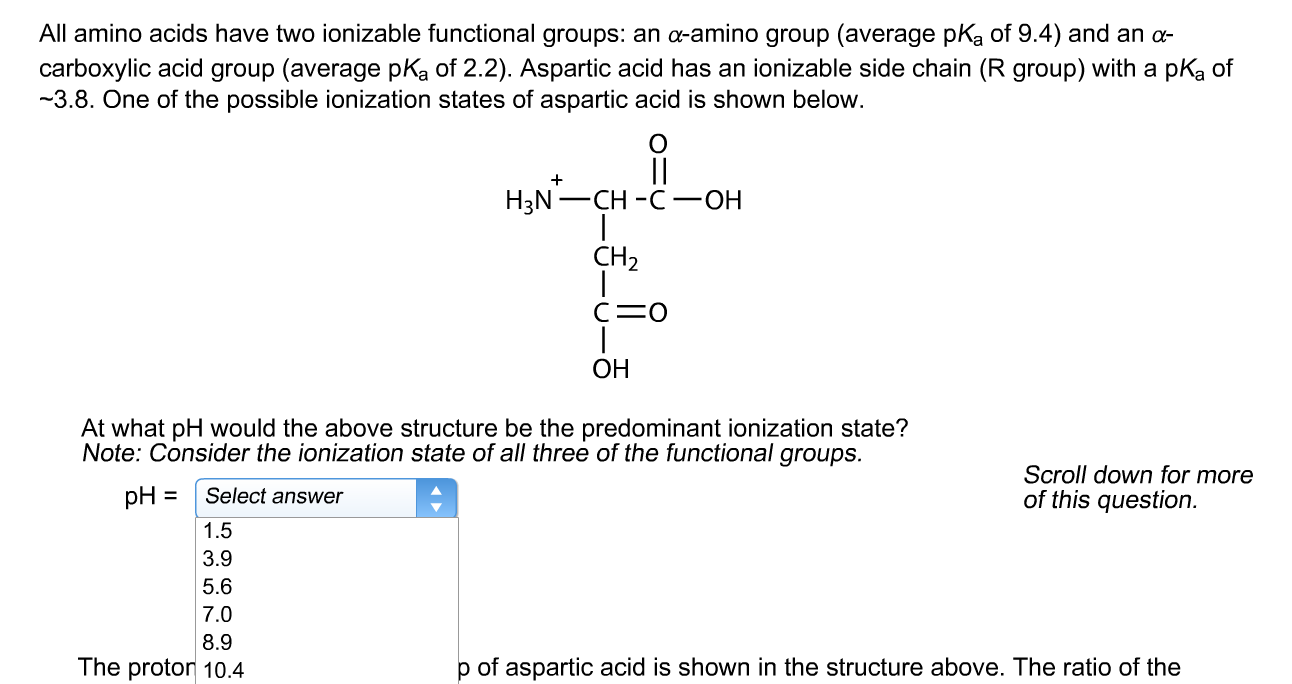
![The \\[pKa\\] values for the three ionizable groups X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point for the amino acid is:\n \n \n \n \n (A)-7.00(B)-3.25(C )- The \\[pKa\\] values for the three ionizable groups X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point for the amino acid is:\n \n \n \n \n (A)-7.00(B)-3.25(C )-](https://www.vedantu.com/question-sets/fd71eb03-6e4b-4038-9de0-4e78ab76280c7204254183704236073.png)
The \\[pKa\\] values for the three ionizable groups X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point for the amino acid is:\n \n \n \n \n (A)-7.00(B)-3.25(C )-

The pK_a values the three ionizable X, Y and Z of glutamic acid 4.3, 9.7 and 2.2 respectively.The isoelectric point the amino acid is:3.257.004.955.95
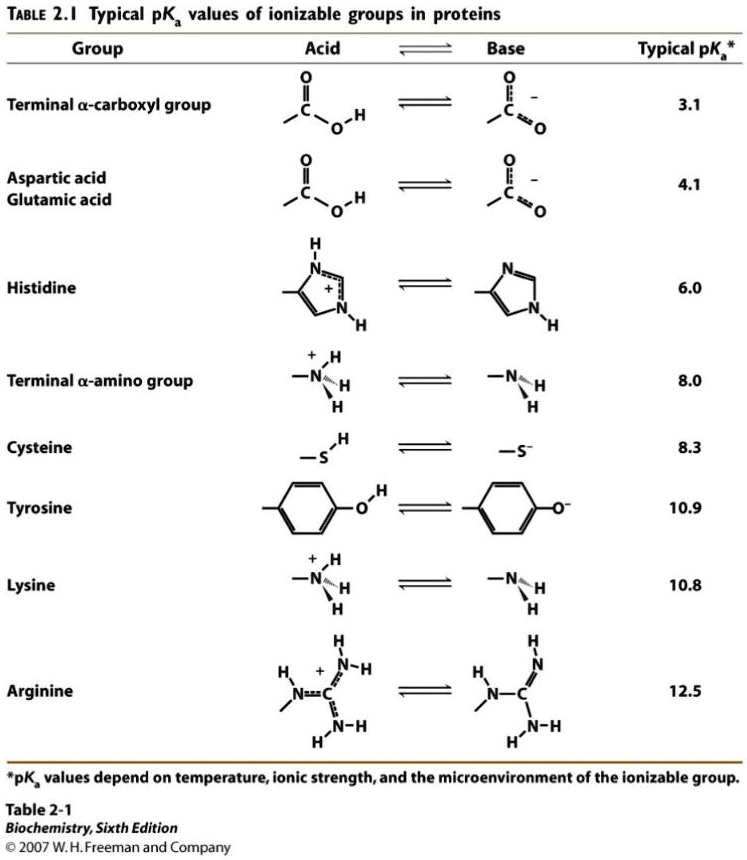
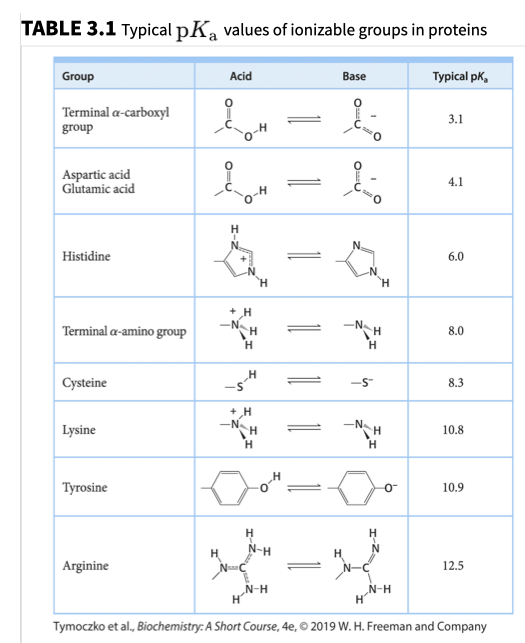
SOLVED: Using the attached pKa table, calculate the isoelectric point of this peptide. TABLE 2.I Typical pKa values of ionizable groups in proteins Group Acid Base Typical pKa* Terminal α-carboxyl group 3.1

All amino acids have two ionizable groups (an alpha-amino group with pKa∼9.3, and an alpha-carboxyl group with pKaα2.2). Lysine also has an ionizable side-chain (R) with a pKa of 10.5. What percentage

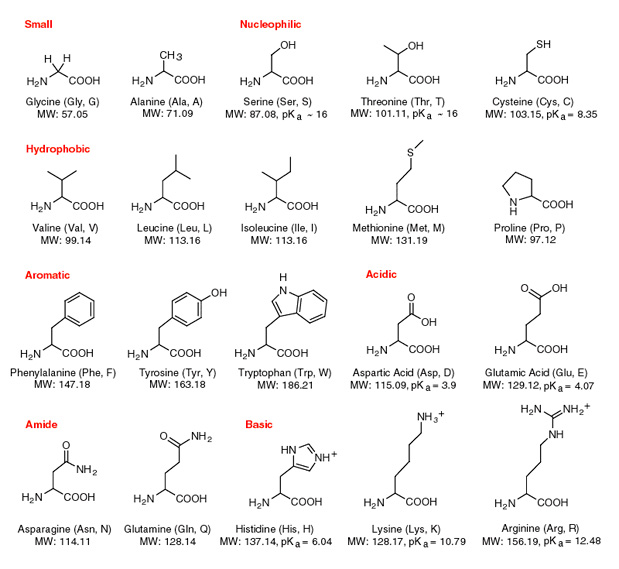
Proteins: Amino Acid Chains DNA Polymerase from E. coli Standard amino acid backbone: Carboxylic acid group, amino group, the alpha hydrogen and an R group. - ppt download

All amino acids have two ionizable groups (an alpha-amino group with pKa = 9.3, and an alpha-carboxyl group with pKa = 2.2). Lysine also has an ionizable side-chain (R) with a pKa

Factors influencing the p K values of ionizable groups in proteins. A ,... | Download Scientific Diagram

Extension of the SMARTS list of ionizable groups. (A) Illustration of... | Download Scientific Diagram

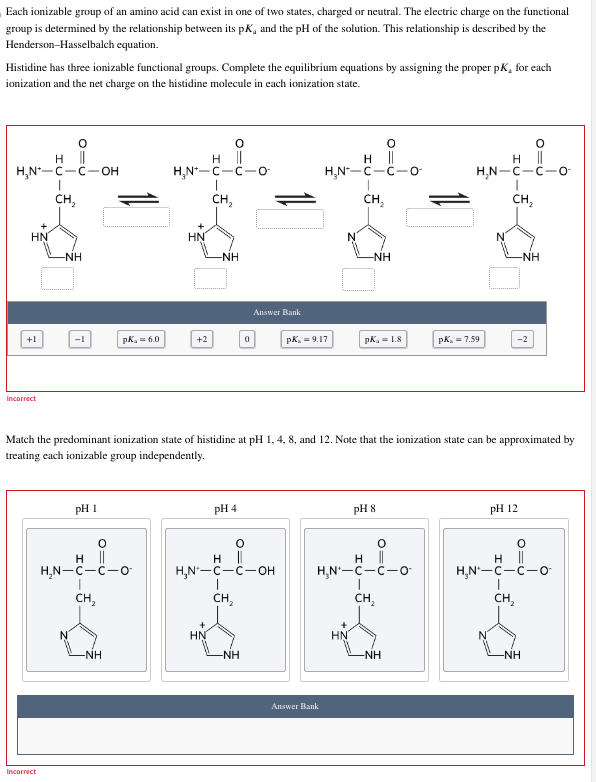
SOLVED: The pK for all the ionizable groups within histidine are labeled accordingly. What will the net charge for this amino acid be in a solution at pH = 7? 1.82 9.17
![Ionizable groups of hen egg white lyzosyme (HEWL) [16]. The inidividual... | Download Scientific Diagram Ionizable groups of hen egg white lyzosyme (HEWL) [16]. The inidividual... | Download Scientific Diagram](https://www.researchgate.net/publication/279384100/figure/fig3/AS:669055061090313@1536526565249/Ionizable-groups-of-hen-egg-white-lyzosyme-HEWL-16-The-inidividual-amino-acids-are.png)